What is the ERA Test?
- The Endometrial Receptivity Analysis (ERA), developed and patented by Igenomix (PCT/ ES2009/000386), is a test designed to evaluate endometrial receptivity.
- The ERA test has been shown to be highly sensitive and specific in the detection of genetic expression profiles associated with receptivity. It allows a patient’s personalized window of implantation to be detected and applied to their future frozen embryo transfers.
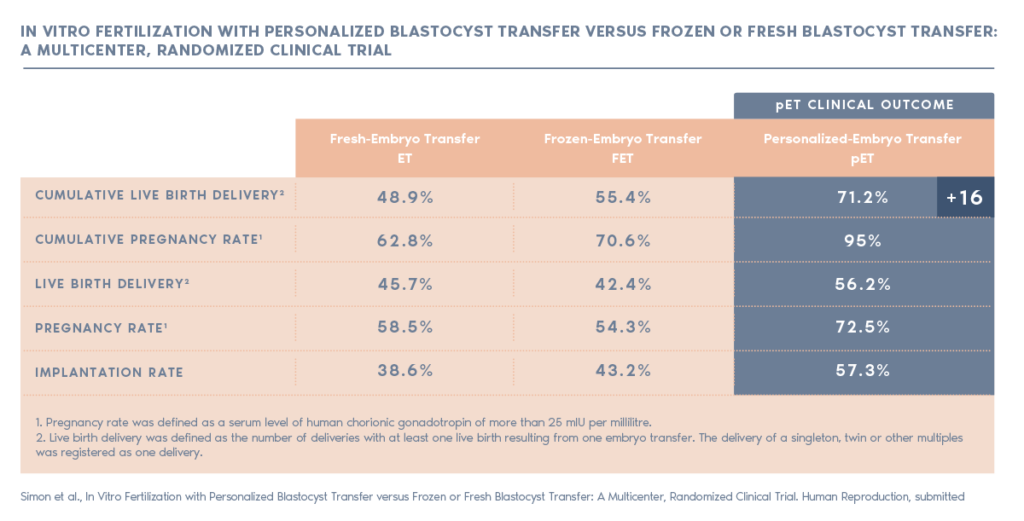
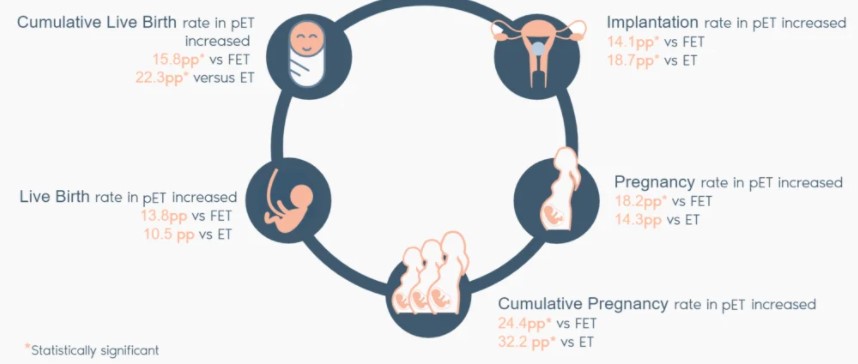
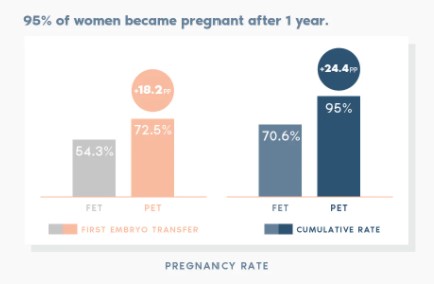
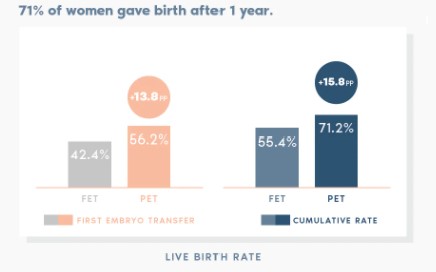
ERA RCT CONCLUSIONS
Our randomised study shows that customising embryo transfer to the day of the cycle on which the patient is receptive has better results than transferring on a standard day.
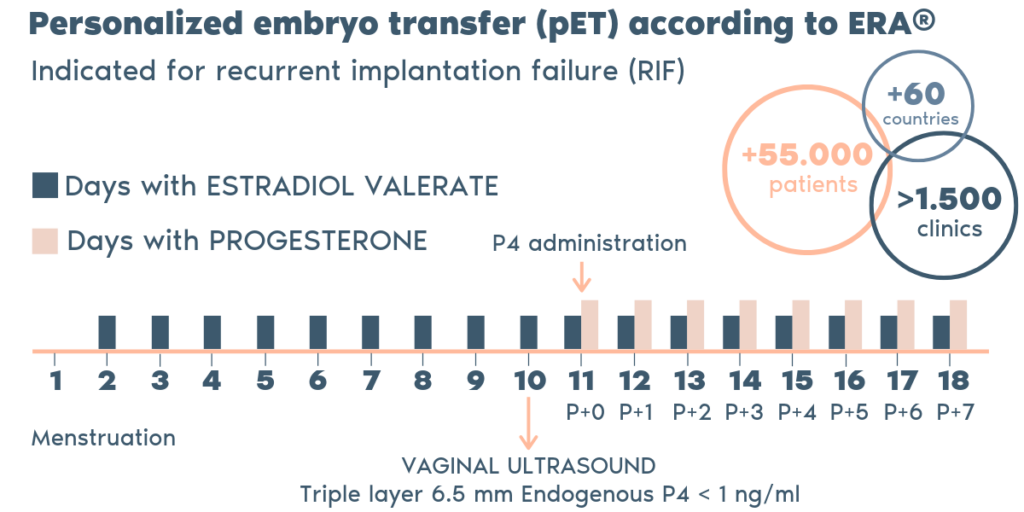
What is the procedure?
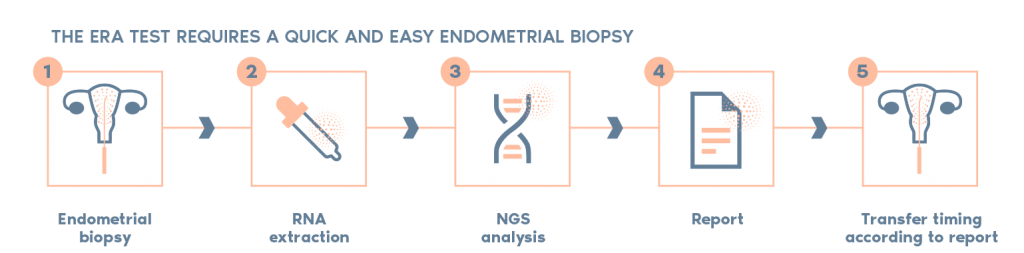
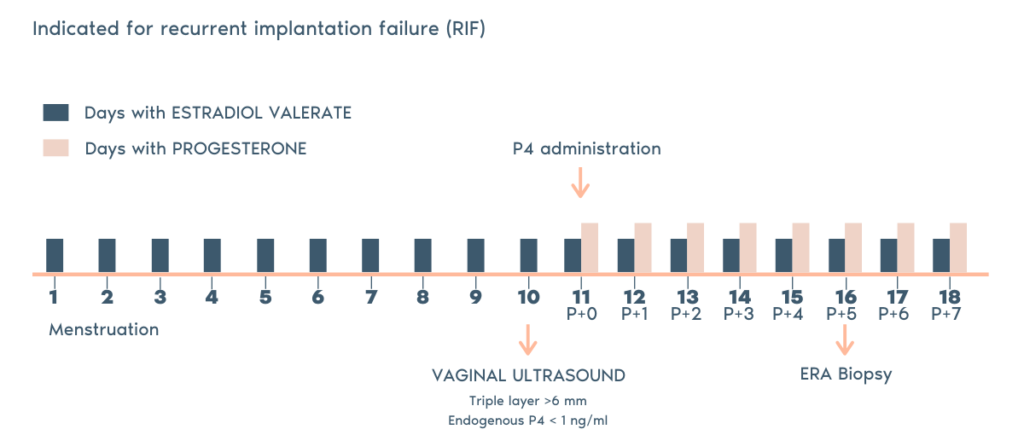
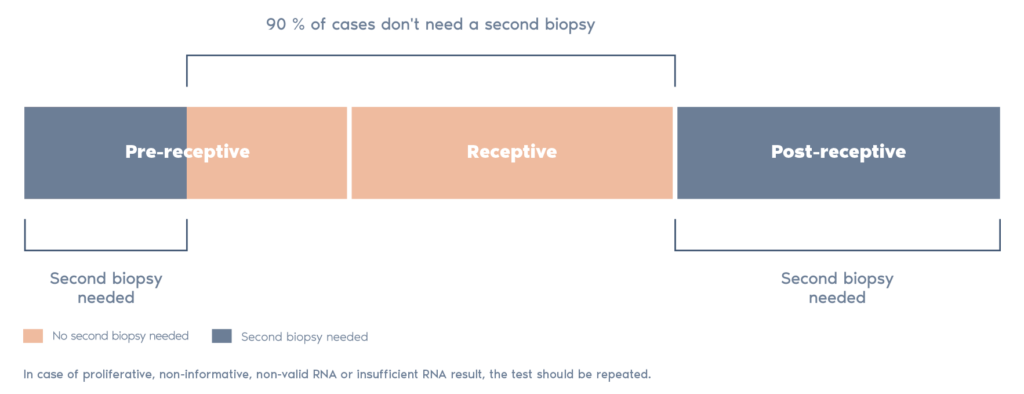
- The ERA utilizes RNA obtained from an endometrial tissue sample. The RNA analyzed and then classified by our ERA predictor as receptive or non-receptive, depending on the expression profile of the RNA.
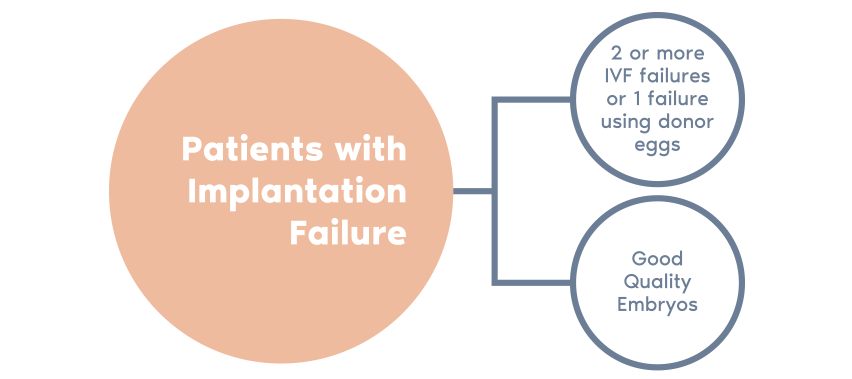
- If the endometrium is non-receptive, the test enables us to find a personalized window of implantation for each patient in the majority of cases.